Resistance in GIST warrants a different therapeutic approach1
Advanced GIST is a highly complex disease with a myriad of mutations fueling resistance and progression1,2
GIST is often driven by primary, activating mutations in kinase genes.3
- These mutations keep the kinase in the active state, causing uncontrolled cell proliferation and/or cell survival4,5
| Molecular Classification of GIST3,6,7 | ||
|---|---|---|
| Gene/exon | Primary mutation frequency |
Secondary mutation frequency |
| KIT | 70-80% | |
| Exon 9 | 10% | |
| Exon 11 | 60-70% | |
| Exon 13 | 1% | 56% |
| Exon 17 | 1% | 41% |
| PDGFRA | 5-10% | |
| Exon 12 | 1% | |
| Exon 14 | <1% | |
| Exon 18 (D842V and other) | 6% | 3% |
| Wild type SDH Deficient, NF-1, BRAF |
10-15% | |
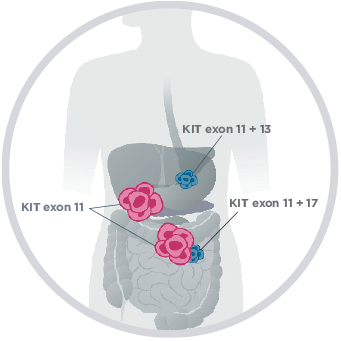
Advanced GIST is marked by the development of secondary, drug-resistant mutations, which play a key role in disease progression2,3,8
- Broad inter- and intra-tumor heterogeneity exists among secondary resistance mutations
A single patient with GIST may have multiple mutations WITHIN or BETWEEN tumors1,9
![]() Primary lesion
Primary lesion
![]() Secondary lesion
Secondary lesion
For illustrative purposes

The majority of advanced GIST patients will eventually develop
resistance, and require lifelong TKI therapy10
As resistance develops, treatments may lose their ability to control disease progression
- The vast majority of patients who benefit from front-line TKI therapy will eventually develop resistance2,3
- As secondary resistance mutations emerge, some TKIs may lose their ability to prevent kinase activation, leading to cancer cell proliferation6,9,11

Currently there are several therapies for advanced GIST patients, yet some patients still do not receive all treatments they are eligible for
Life‑long systemic therapy is recommended by the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for TKI‑sensitive recurrent or metastatic GIST12
Clinical experience suggests that discontinuing TKI therapy, even in the
setting of progressive disease, may accelerate the pace of disease progression and worsen symptoms12
Median PFS and ORR13-16
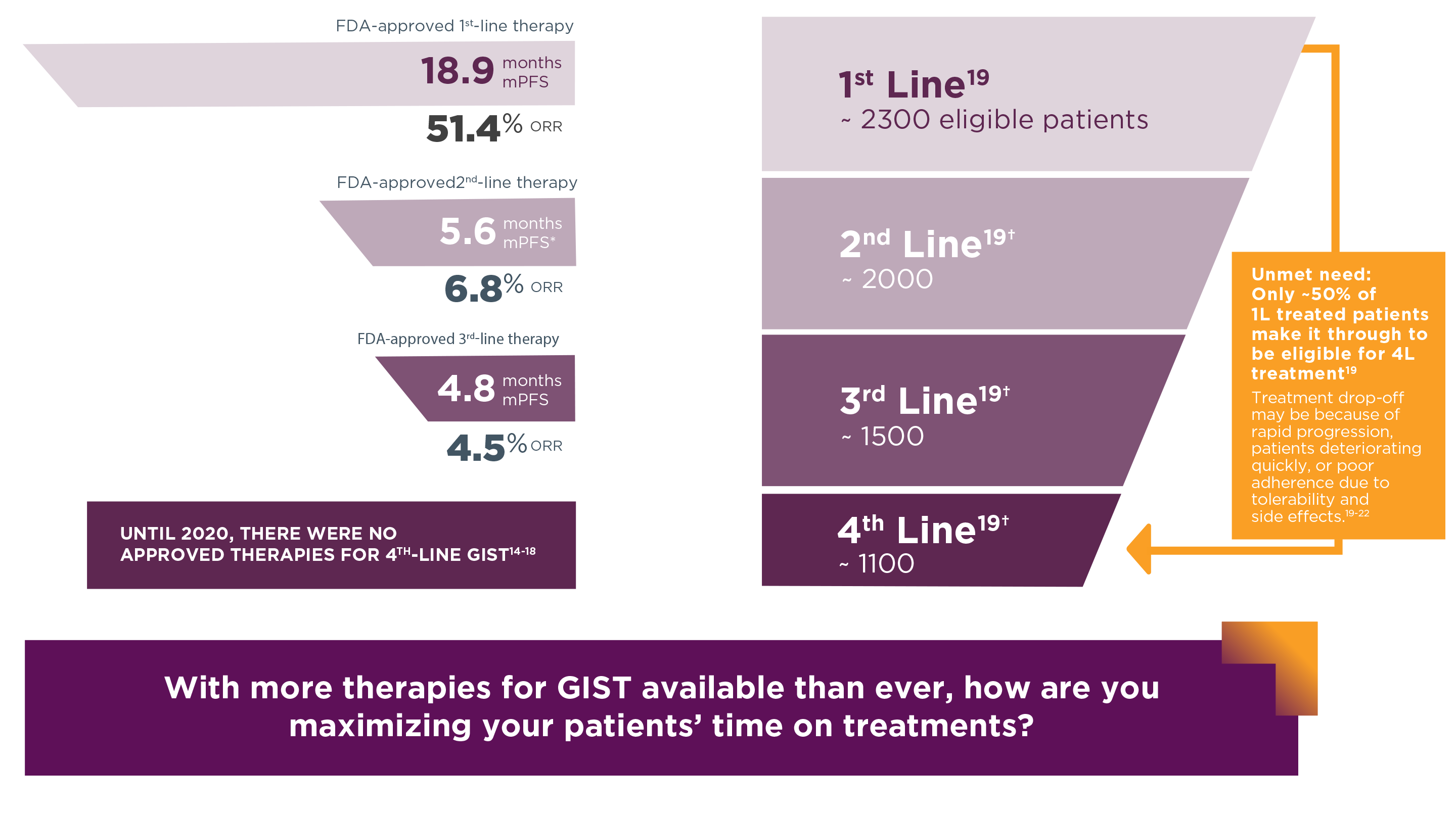
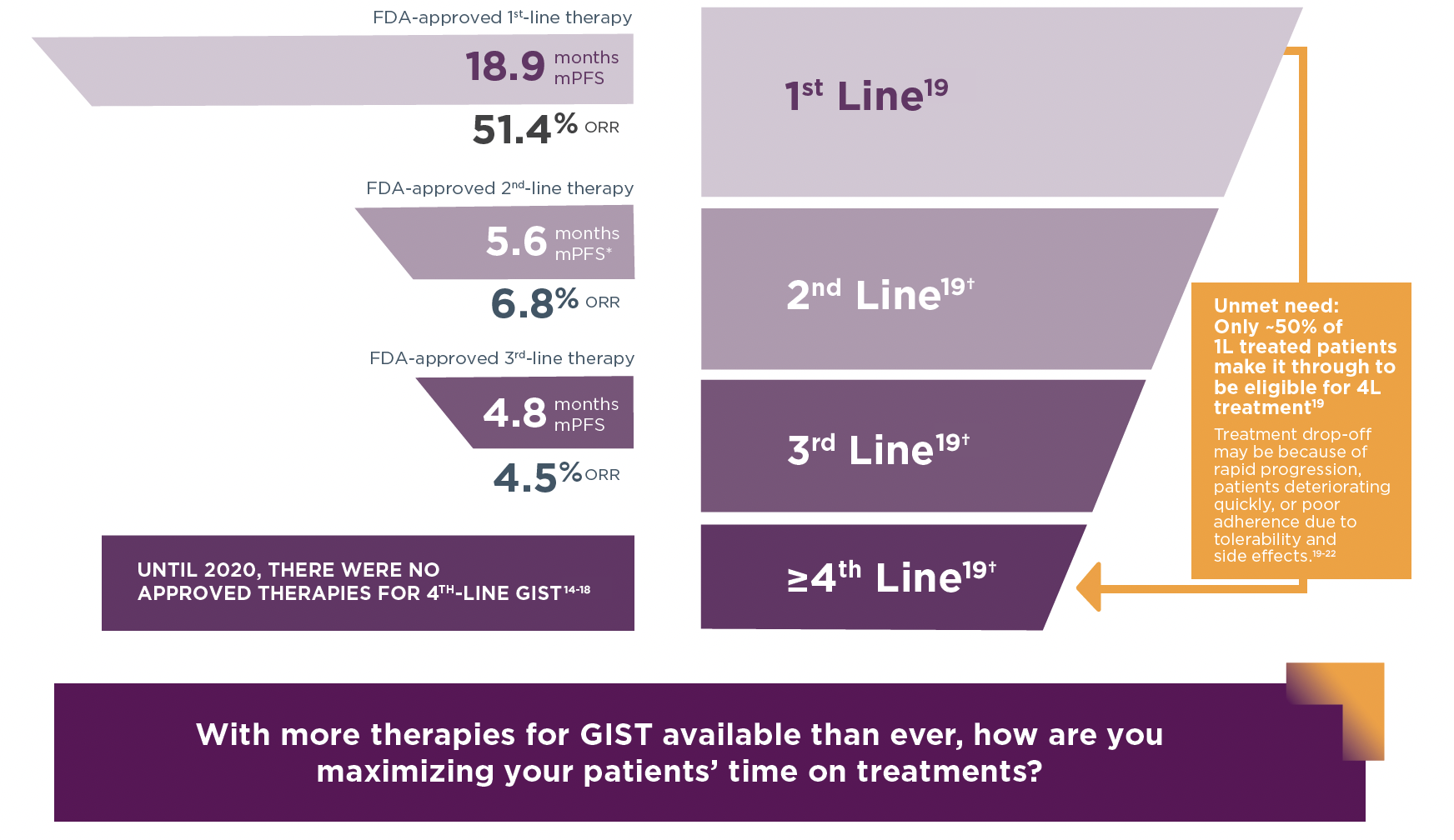
- The FDA-approved 1st-, 2nd-, and 3rd-line therapies for GIST are imatinib, sunitinib, and regorafenib, respectively14-16
- Avapritinib is approved for patients with unresectable or metastatic GIST harboring a PDGFRA exon 18 mutation (5–6% of patients), irrespective of line of therapy3,17
*PFS converted from weeks to months.
† Excludes estimated proportion of patients who are not eligible for treatment due to death, discontinuation of oncology treatment, or clinical trial enrollment based on recent analyses of US claims data.
Qinlock is engineered to block the drivers of resistance in advanced GIST11,18
Qinlock is the first and only switch-control kinase inhibitor that broadly inhibited KIT and PDGFRA kinase signaling in preclinical studies11,18
Powerful PFS Results
Qinlock significantly improved PFS vs placebo (6.3 months vs 1.0 month; P<0.0001)18
See The DataRobust Phase 3 Trial
The efficacy and safety of Qinlock was
demonstrated in the INVICTUS trial18